For centuries, we humans are fighting contagion, working hard to catch, treat or prevent disease, but history has shown us that success has been limited, short-term, and any progress met with an arrival of a new disease – sometimes the same enemy in a new form. The vaccine has remained our most powerful weapon against many lethal pathogens whose development has a history of its own.
This war, however, still continues as only one battle has truly been won to date, against smallpox, an ancient virus once feared throughout the world. It was with us for at least 3,000 years back to the Egyptian era.
Dangerous smallpox and suffering of humanity what could we do?
The smallpox was very deadly and was even called the ‘the scourge of mankind’, making it a priority to protect us, humans, against this really bad disease.
The virus that causes smallpox — variola — attacked almost anyone it came into contact with, starting with a fever followed by a unique rash. Fluid-filled spots containing the virus would soon take over the whole body, bringing death to many and leaving survivors scarred for their lifetime.
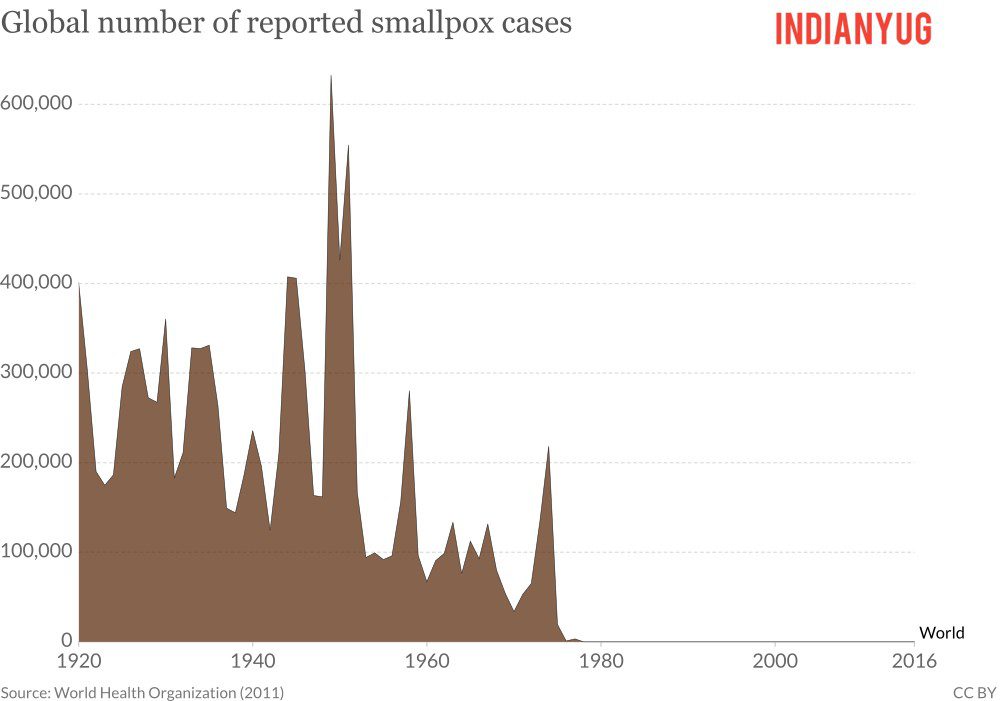
Smallpox alone killed 300 million people in the twentieth century. When it was removed from our population, its end is regarded to be the biggest achievement in international public health by far.
But most experts know how challenging a similar victory will be. Some say it is most likely impossible, as smallpox was simply a perfect target to be eradicated. Multiple efforts beforehand had failed — first against hookworm, then yaws, then malaria, and current efforts to end two other diseases — polio and Guinea worm — are lagging decades behind.
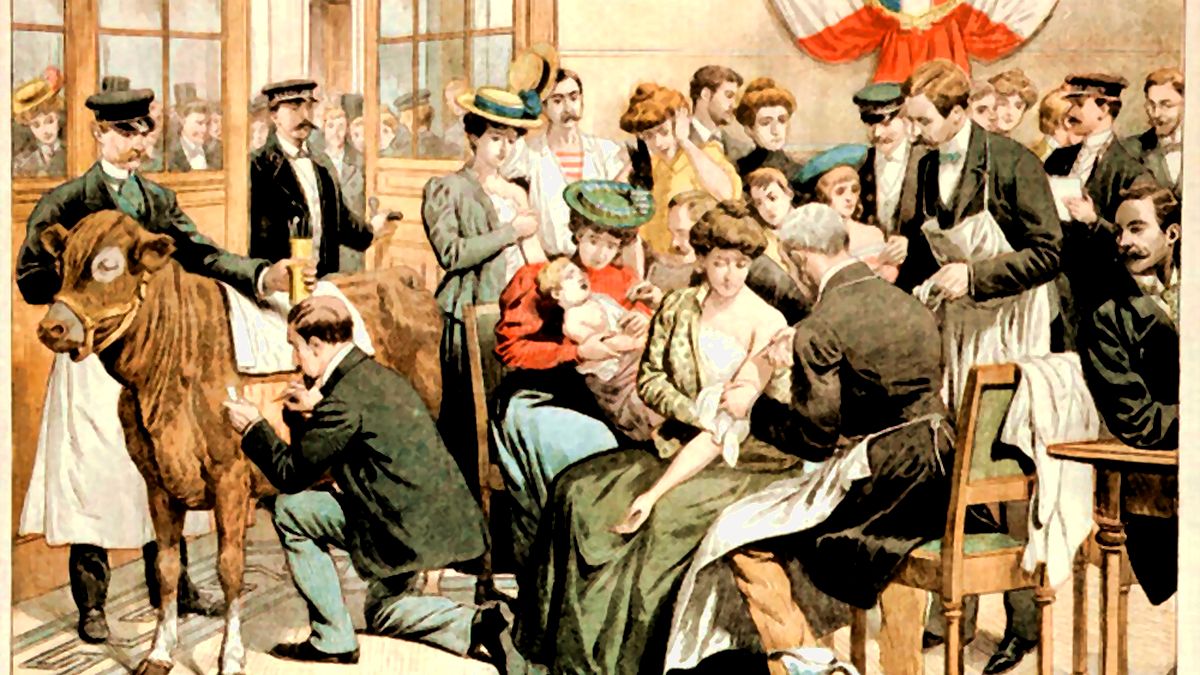
Cow and the beginning of vaccination that will save millions of lives in time to come

Smallpox killed approximately three out of every ten infected while sparing many with lifelong scars. As we know, the world was officially rid of smallpox in 1980 after an eradication effort that had begun thirteen years prior. However, there is a history and story behind the success in eradicating the deadly disease which otherwise could have killed millions.
The story that led to the end of the disease began almost 200 years earlier, on the arm of an eight-year-old boy named James Phipps. Blossom the cow, was also the central character of the story, which brought the first vaccine in the arms of James Phipps.
Phipps was the son of a gardener who just happened to work for Edward Jenner, an English physician, and scientist who would later come to be known as the father of immunology.
For years, Jenner had heard rumors that milkmaids exposed to cowpox become naturally protected against smallpox. The cowpox virus closely resembles variola and word had spread that humans who came into contact with cowpox developed a milder disease they soon recovered from, which left them immune to its more lethal relative, smallpox.
While such experimentation can’t even be imagined today, Jenner decided to test the theory that cowpox could be given deliberately to humans as a means of protection — and Phipps would be his proof.
In May 1796, Jenner found milkmaid Sarah Nelmes, a recent cowpox patient. Nelmes apparently caught the virus from a cow called Blossom (whose horn is now on display in Jenner’s house in Berkeley, Gloucestershire; her hide is kept in the St George’s Medical School library in Tooting, south London).

Jenner sampled the virus hiding in Nelmes’s lesions and used it to inoculate young Phipps. The eight-year-old young boy went on to develop a mild fever and loss of appetite but recovered after ten days.
Two months later, in July, Jenner exposed Phipps to smallpox and no symptoms or lesions developed. He seemed to be protected. Jenner went on to successfully repeat this on more people over the following two years, again poor laborers, their children, or inmates of workhouses.
In the following years, however, people across all classes were inoculated. The world vaccination (after the Latin vecca for ‘cow’) soon became universally accepted. The groundbreaking idea that our bodies could be protected against infection, using an infection, was born and smallpox would eventually become the target of a global defense, creating a biological shield that would one day span across the planet. But the road to get there would be far from straightforward.
You may also like: 1918 Spanish Flu in India is a Lesson in Fighting Coronavirus Outbreak Now
Ending smallpox with vaccine
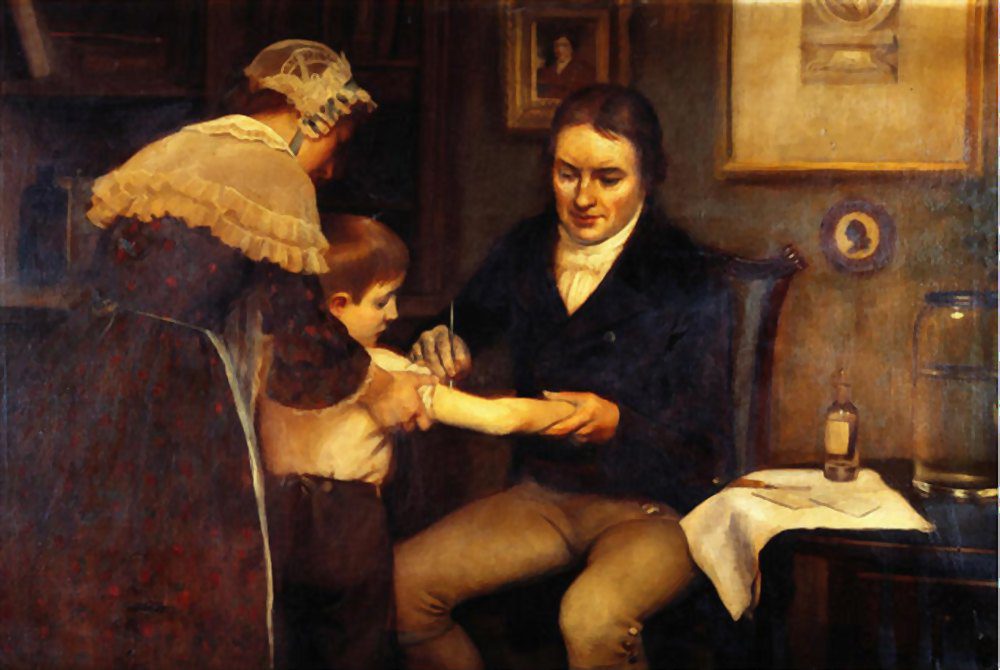
A full efforts program to eradicate smallpox began in 1967 when there were still more than 10 million cases occurring across 43 countries. By this point, the disease had already been eliminated — meaning it had stopped spreading in a particular geographical region — in North America and Europe, following an initial effort to end the disease, launched by the World Health Organization (WHO), in 1959.
The program had focused on vaccinating the masses and the target had been to get at least 80 percent vaccine coverage in every country in order to reach the herd immunity threshold, a level of coverage where the chances of unvaccinated people getting the disease are extremely low.
The threshold for herd immunity varies from one disease to another, based on how easily the infection transmits between people.) But South America, Asia, and Africa continued to see millions of cases, while Europe and North America were still seeing imported cases, particularly as air travel rose in popularity.
The birth of surveillance-based approach that has since become the backbone of outbreak control

Dr. Donald A. Henderson, who died in 2016 at the age of 87, had led the international effort to end smallpox in the middle of widespread criticism that it was an impossible task.
He was right in his initial assessment that simply vaccinating everyone against smallpox, or any disease was not always feasible and therefore should not be the only strategy.
Public health teams needed to understand the severity of the situation – how many were affected and where — to better target their resources as well as to contain those infected to stop them from spreading the disease further. ‘We made a very strong point about the need for surveillance of cases and their containment,’ he said. Epidemiologist Dr. William Foege soon implemented a surveillance and containment strategy under Henderson’s leadership and significant reductions were quickly accomplished.
For example, Foege’s team used limited resources to focus solely on outbreak-affected areas when working in eastern Nigeria in 1967, identifying cases and vaccinating everyone within a defined radius of an outbreak, known as ring vaccination.
This halted the outbreak within five months, despite only 750,000 people of the 12 million people living in the region receiving the vaccine. The method was proven to work again and again and was simply much more efficient than trying to reach everyone.
The disease usually affected 5 percent of a population at most at any one time, so the aim was to identify those 5 percent infected and focus efforts there. Using this approach, Hopkins’ team saw results within months in Sierra Leone, and within a few years in the West African region as a whole, despite the region having the worst infrastructure of any they were working in globally.
You may also like: Here’s Why It’s Taking So Long to Develop a Vaccine For COVID-19
Difficult situation in India

The situation was much more difficult in populous India, Hopkins notes, as four years after success in West Africa, teams were still failing to see a significant improvement.
Government teams set out to visit every household in the country in a period of ten days in 1973 to know the true extent of cases and stop the disease spreading more quickly.
Soon the true extent of the spread became obvious, some states were found to have twenty times more cases than previously reported. However, once this was known, resources were quickly deployed with greater accuracy, and India reported its last case of smallpox just one year later.
This surveillance-based approach has since become the backbone of outbreak control.
The concept was simple, that if we could discover the cases more quickly than before, the containment teams could interrupt the chains of transmission, teams could then break those chains by vaccinating possible contacts in areas where there were cases.
Additional factors further helped the success of the global eradication campaign — for example, community teams ventured out to people rather than waiting for them to come to health facilities, meaning they reached everyone, including the most remote.
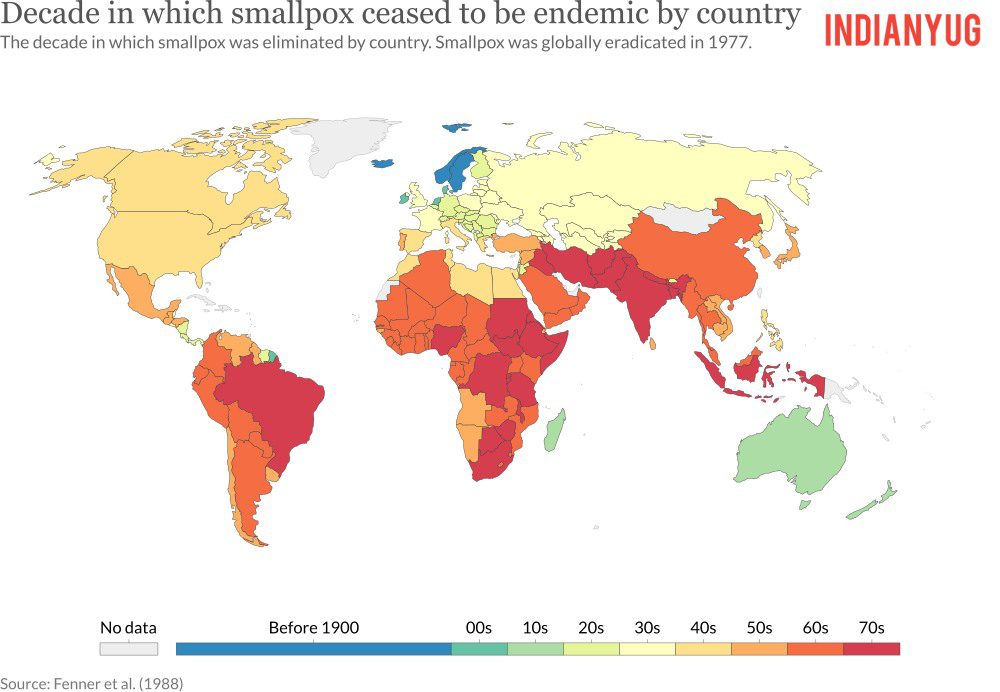
A further development was the introduction of a bifurcated needle in 1968, a thin metal rod with two prongs that would hold a dose of the vaccine for more efficient injection into the skin.
The beauty of this ingenious tool was its simplicity when compared to the jet injectors previously used; it enabled 100 doses to be delivered from a single vial. But despite these factors coming together to bring immediate progress in reducing cases, the road to eradication took twelve years, with societal and political elements coming into play, such as civil wars, extreme weather, poor infrastructure, and, importantly, convincing people the vaccine was safe.
As these difficulties were overcome and cases of smallpox decreased, the need for resources in order to find the remaining cases increased. The cost per case rose significantly as teams traveled farther and wider to find the final few.
But they did find them. On 26 October 1977, experts found and isolated the last case of naturally occurring smallpox in 23-year-old hospital cook Ali Maow Maalin. Maalin, from Merca, Somalia, had worked as a vaccinator in the smallpox program, yet had avoided the vaccine himself due to a fear of needles.
The virus caught up with him as he helped direct a driver taking two children to a nearby smallpox isolation camp. Nine days later Maalin developed symptoms. He was told to stay home while special teams were sent to vaccinate the households around his location, successfully reaching more than 54,000 people in two weeks.
Maalin recovered and smallpox was over. Almost. Janet Parker, a medical photographer, and her mother were the last official cases of smallpox, in August 1978.
Parker is presumed to have contracted the virus at Birmingham University medical school, where research on the virus was taking place. She became ill on 11 August and developed the signature rash on the 15th, but was not diagnosed with smallpox until nine days later.
She died on 11 September. Her mother, who had been caring for her, also contracted the disease but survived. Parker had access to the laboratory where a smallpox specimen was contained, but it is unclear if she was infected by entering that laboratory or by the ventilation system taking the virus from the laboratory to her office in the same block, explains David Heymann, Professor of Infectious Disease Epidemiology at the London School of Hygiene and Tropical Medicine, who worked with the eradication program for two years.
You may also like: Indian Coronavirus Mutation Now Threatens Vaccine Development for COVID-19
Voices for destruction of specimens of virus
The Birmingham University event prompted a program of consolidation or destruction of the remaining specimens of the virus. This took place during the Cold War, and countries were given the choice of providing their smallpox stocks to the United States, to the USSR, or destroying their stocks under a set protocol.
Both the US and Russia continue to hold on to the stocks given to them, monitored and handled by the World Health Organization under a WHO agreement set in 1979. The US stock is held at the Centers for Disease Control and Prevention (CDC) in Atlanta and the Russian stock at a research laboratory in Siberia.
The two facilities are inspected by the WHO every two years. Debates on whether these last remaining stocks of smallpox should be destroyed have been ongoing for decades. Research on the virus continues today, following anthrax attacks in the United States in 2001, during which anthrax spores were found lacing mail sent to news agencies and congressional offices.
This led to the development of bioterrorism preparedness programs, which include research on smallpox, looking for better diagnostic tests, antiviral medications, and safer vaccines. But gene technology has enabled the smallpox virus to be fully sequenced, meaning that the virus can be reconstructed for research purposes if needed, which some experts, including Heymann, believe removes the need for live virus stocks to be stored.
The World Health Assembly, the decision-making body of the WHO, has requested the review of research using the live smallpox virus on multiple occasions, with the 69th assembly calling upon an Advisory Committee to review this in May 2016.
At the 72nd assembly in 2019, the committee stated that research using the virus was still needed to continue the development of antiviral medicines for smallpox preparedness, so the stocks remain. Officially, though, smallpox has been eradicated, with Ali Maow Maalin being the last face of the smallpox pandemic that plagued the world for millennia.
The eradication laid the groundwork for the routine vaccination programs now implemented globally, in the WHO’s Expanded Programme on Immunization, protecting children from multiple childhood diseases including measles, polio, and tetanus.
Could these too be eradicated? Most experts say no, but polio efforts are underway. ‘Even towards the end of smallpox eradication, the senior staff never talked about potential eradication of any other disease,’ Henderson told the WHO.
We had many things on our side in our fight against smallpox — the vaccine was heat stable and did not require refrigeration for storage, an invaluable property in the remote, tropical settings in which the vaccine was used.
Furthermore, immunization required just a single vaccine dose, and everyone who had the disease could be identified by its distinctive rash and therefore easily isolated and their contacts vaccinated.
Other diseases do not have this combination of winning elements; some have just a few, and most only one or two.
But as pandemics and global health emergencies increase in frequency, how can teams not hope to at least try to rid the world of some of them for good?